The first patient arrived just over two years ago. January was supposed to be a slow month at Santa Rosa, a hospital nestled in the middle-class Pueblo Libre district of Lima, Peru. The sprawling metropolis of 10 million can feel eerily empty at the height of summer, when some families flee the city and many others flock to the beach.
The patient, a woman in her early 20s, had traveled 270 miles with her mother from their home in the central Peruvian jungle. She had a high fever and unbearable stomach pain — the result, she feared, of a recent abortion gone wrong. The doctors at Santa Rosa took X-rays and found blood moving freely in her abdominal cavity. Acting quickly in a desperate bid to stop the bleeding, they decided to remove her uterus.
But the hemorrhage continued. The doctors ordered every test they could think of. Just one came back positive, and it was something most of the physicians had never encountered. The patient was not experiencing pregnancy- or abortion-related complications after all; instead, she was in the most severe phase of dengue fever, a disease rarely seen in Lima at the time due to its temperate climate. In any other year, the case would have been an outlier. After all, the patient was not local and had come from the Amazon, where mosquito-borne illnesses like dengue are perennial risks.
But this time, it was the beginning of an epidemic.

Zoya Teirstein / Grist
Dengue was once a hazard throughout the Americas, but pesticide-spraying campaigns appeared to vanquish the disease by the mid-1900s. Peru declared the Aedes aegypti mosquito — the main carrier of dengue — eradicated in 1958. But with an outbreak in 1990, the worm began to turn, in Peru and around the world. Now, the number of dengue fever cases reported globally has been growing for decades, from 500,000 cases reported to the World Health Organization in 2000 to over 5 million in 2019.
While improved disease reporting accounts for part of this tenfold increase, experts primarily blame urbanization — which creates ideal conditions for the Aedes mosquito by providing ample pockets of water for it to breed in and dense human populations to bite once hatched — and the explosion of air travel, which enables the disease to escape endemic zones. As a result, dengue is the most common and fastest-growing mosquito-borne disease in the world.
But something else has happened in just the past few years. In 2023, the global case count approached 7 million — a 40 percent jump in a single year. And even that new record was crushed in 2024, when 14 million dengue cases and 9,000 deaths were reported worldwide, the majority of them in the Americas.
“It’s different from earlier years when dengue was around but you don’t see it much,” said Luciano Andrade Moreira, an agricultural engineer and entomologist in southern Brazil. Seventeen cities in Brazil declared states of emergency. Hospitals overflowed. So many people were sick that the crisis began to assume the character of society-wide disorder, similar to that experienced during the recent COVID-19 pandemic. Some supermarkets didn’t have enough cashiers to staff checkouts. “You see the disease coming next to you,” said Moreira, whose brother and sister-in-law were both infected.

Mateus Bonomi / Anadolu via Getty Images
In Lima’s Santa Rosa hospital, surgeon Yolanda Sánchez remembers that the first dengue patients “came like a wave.” No sooner was the young woman who unknowingly became patient zero discharged than her bed was immediately filled with another dengue patient, and another after that. Some hallways were so crowded with patients that staff had trouble navigating through the throng. The sick filled every available side room, spilled out onto the street, and massed near the wrought-iron front gate.
At the peak of the outbreak in February and March, Santa Rosa was fielding between 40 and 60 dengue patients a day — an unprecedented onslaught for a facility that saw just 13 such patients in all of 2022. The sick looked like a random sampling of the Peruvian population: young, old, chronically sick, and otherwise perfectly healthy.
When a woman under 60 with no comorbidities entered the hospital and died less than 24 hours later, a quiet doctor named Solomon Durand knew Santa Rosa would need to adapt if it was going to survive the epidemic. Durand, an epidemiologist, had recently come to Santa Rosa after a 15-year stint at a hospital in Iquitos, a northern Amazonian city long a hotspot for mosquito-borne disease. He was the only doctor at Santa Rosa who had worked through dengue outbreaks before, and he knew what to do.
Durand set up a tent in the hospital’s back parking lot to quickly assess new patients. Those with severe dengue were moved into an intensive care unit for monitoring. Everyone else was sorted into different wings of the hospital, treated, and released as quickly as possible. Durand monitored the files of the admitted patients; what struck him was the proximity of the outbreak. More and more of Santa Rosa’s dengue patients were coming from Pueblo Libre and the six other districts surrounding the hospital. These neighborhoods didn’t suffer from the overcrowding and poor sanitary conditions that usually spread dengue in urban areas, including in the limited outbreaks Lima had weathered in the past. “That caught our attention,” Durand told me. He knew something else must have shifted.


A privacy screen partially blocks the doorway of a room at Santa Rosa hospital in Lima, Peru. Nearby, a hospital worker wheels supplies between rooms. At the peak of the 2024 outbreak, the hospital was seeing up to 60 new dengue patients per day. Zoya Teirstein / Grist
The next clue occurred to him as the outbreak stretched on through an exceptionally hot summer and into the fall. A year earlier, the natural climate pattern known as El Niño had developed in the tropical regions of the Pacific, altering rainfall patterns and raising average temperatures across the globe for the next 12 months. Durand noticed El Niño’s effects in Lima. In the weeks before and after that first dengue patient arrived, unusually intense rains had flooded the city, which is typically in a semi-permanent state of drought, eventually prompting the Peruvian government to declare a state of emergency. The rains then gave way to an unrelenting summer sun.
“That year there was more sunshine in Lima,” Durand recalled. “The summer lasted longer.” It was the country’s warmest year in over six decades.
Durand started to wonder if the unknown factor driving the outbreak was climate change.

Hotter conditions act as an accelerant for dengue fever. Rising temperatures not only boost the development of the Aedes aegypti and Asian tiger mosquitoes that carry the disease, but they also speed up the replication of the dengue virus inside of those insects. In recent years, researchers have sought to isolate the effect of global warming on the spread of the disease. In September 2025, scientists at the University of Washington determined that, in the 21 countries across Asia and the Americas with the highest exposure to dengue, 18 percent of cases between 1995 and 2014 would not have occurred in the absence of human-caused climate change. It’s the first direct scientific evidence linking global warming to dengue’s widening global footprint — confirming the hunch Durand had more than a year earlier, when he noticed dengue’s spread into what had been a formerly inhospitable environment.
The problem is poised to get worse. By midcentury, the authors found, climate change could lead to an additional 50 percent increase in cases in the countries they studied. And that’s if new greenhouse gas emissions are kept low enough to keep warming below 2 degrees Celsius (3.6 degrees Fahrenheit).
If there were a powerful, affordable vaccine against dengue, the accelerating growth in caseloads wouldn’t be nearly as daunting. More than 30 years of research and work on finding a vaccine that simultaneously protects against all four dengue serotypes in circulation has yielded limited success, though Brazil recently approved a single-dose vaccine that looks promising. More than a million doses of the vaccine are currently being distributed to a handful of pilot cities, so the world may soon know if a viable vaccine is on the horizon.
In the meantime, about half the world’s population is now at risk of dengue.
In Lima, the 2024 epidemic hit 40 of the city’s 43 districts simultaneously. But thanks to Durand’s quick thinking and prior experience, only four of the nearly 2,000 diagnosed patients at Santa Rosa died. Even after the crisis finally subsided in June, the doctor continued to study what had happened, eventually publishing an article on the outbreak in a health sciences journal. More than two years after the first patient arrived, Durand is still ruminating over how ill-prepared his hospital was for a crisis he thinks will only become more common as climate change worsens.
“If we had even suspected that this was going to happen, we would have prepared ourselves in advance,” he said. “There was no warning.”
Across South America, researchers and public health departments are working furiously to make such a warning possible. The strategy, years in the making, is something of a double-tipped spear. One tip is the use of machine learning to predict outbreaks months in advance. The other enlists the natural world to prevent dengue spread in the first place, releasing hundreds of millions of mosquitoes that have been carefully infected with a dengue-blocking bacteria into cities. When these mosquitoes breed with their wild brethren, they suppress the disease in future generations.
Unlike the reactive pesticide-spraying campaigns that defined vector-borne disease control throughout the 20th century, these programs seek a more deliberate cooperation with existing ecosystems. But they also face substantial logistical and political challenges, requiring public officials to spend money on programs that won’t pay obvious dividends for years — assuming anyone notices their success at all. But as dengue changes shape, health officials in countries like Peru and Brazil are not just noting the value of investing in such an approach, but demanding it.
“Politicians, they want to solve the problem tomorrow,” said Moreira, who is himself spearheading a mosquito-breeding effort in Brazil. “When you suffer, it changes the concept.”
The Iquitos-Nauta highway, which connects the two northern Peruvian cities in its name, was paved in 2004. The bustling highway and the dirt road offshoots it has spawned are lifelines for the extremely isolated communities living in the Peruvian Amazon. Many lack running water or refrigeration, and the new access to medical care and other urban amenities is saving lives.
But in this biodiverse region, the roads have also opened up new opportunities for infectious diseases to take hold. Anopheles mosquitoes, which carry malaria, and dengue-carrying Aedes aegypti fester in disturbed habitats like those of the newly-accessible Peruvian jungle. Every stand of trees felled by a machete to make way for a new home allows the insects to more easily find standing water and feed on humans. Illegal mining and other clandestine activities in the rainforest leave behind trash, deep gashes in the soil, and other places for mosquitoes to breed. Layer climate change on top of this — extreme temperatures in the Amazon increased half a degree C per decade between 1981 and 2023 — and the scale of the public health challenge becomes even more daunting.

But these same factors are also turning the Iquitos-Nauta highway into an important avenue for research, allowing experts to access health and climate data from places that were once impossible to regularly reach. Llanchama, a nearby settlement of fewer than 300 residents on the bank of the Nanay River, is one of 10 local research sites periodically visited by InnovaLab, a disease-research laboratory based out of Cayetano Heredia University in Lima. Gabriel Carrasco-Escobar, a biostatistician who has been working in the Amazon in one capacity or another since his youth, runs the lab with the help of a team of field researchers from both Lima and Iquitos.
As recently as two years ago, Carrasco-Escobar and his team had to board boats in Iquitos and ride for three hours to reach the community, which comprises a school, a soccer field, and a single grassy lane lined with houses. Now, the drive from Iquitos takes an hour, though it’s still another 30 minutes by foot after that. The new accessibility has allowed the researchers to visit much more frequently — and to install a weather station capable of providing real-time, minute-by-minute updates on temperature, humidity, and other factors.

Carrasco-Escobar cautions that the rainforest is a temperamental research partner. The same conditions that make InnovaLab’s expeditions difficult — surprise floods, thick heat, and half-built infrastructure that muddies the border between road and forest — cause mosquitoes to thrive. The insects are tiny, opportunistic, and tend to defy management. There is no one convenient signal that heralds their movements; they’re influenced by wind, rain, warmth, the density of tree roots, and the availability of blood meals, among other factors. That’s especially true of the Aedes aegypti, an enterprising and highly agile day-biting species that is as comfortable indoors as it is outside.
“The mosquito is extremely well-designed — very intelligent, very adaptable,” Carrasco-Escobar said. “All it needs is a tiny chance.”
Carrasco-Escobar and his colleagues want to establish a monitoring system that is just as intelligent as their adversaries. Their aim is to understand the delicate tapestry of environmental threads that pull the bugs from place to place and cause them to proliferate. This means, first and foremost, understanding in real time how humans themselves are changing the environment. To do this, the researchers are deploying not just weather stations like the one in Llanchama but also acoustic sensors affixed to tree trunks to identify birds in flight and people walking through the jungle, air quality sensors to detect pollutants like brush smoke that indicate deforestation, and drone surveys that use infrared radiation to spot movement and new infrastructure on the forest floor.

Zoya Teirstein / Grist

Using sensors, drones, and weather stations, Gabriel Carrasco-Escobar and his colleagues have established an early warning system that remote communities like Llanchama will be able to use to prepare for infectious diseases.
Zoya Teirstein / Grist

These tools, in sum, represent the full spectrum of humanity’s ability to understand some of the deadliest animals in the world — both the Aedes aegypti and the people who make their spread possible. The information they capture is paired with public health data from remote communities and then fed into machine learning systems. The patterns the program identifies comprise the backbone of InnovaLab’s early warning system, which can now successfully predict a dengue outbreak three months in advance.
The surveillance system could permanently alter the region’s relationship with infectious diseases — and, if adopted throughout the rest of Peru, stand guard against future outbreaks in places like Lima. A three-month heads up allows health departments to move resources to areas where they will soon be in highest demand. Hospitals can train staff to recognize dengue and set up triage rooms before they’re needed. Municipalities can advise residents to stay indoors when possible and wear insect repellent outside. These relatively simple, low-cost interventions would have averted much of the suffering Durand saw at Santa Rosa in 2024.
InnovaLab’s work is part of a larger project called Harmonize, which is named for the integration of climate, health, and ecosystem data central to the work. (Harmonize is a project funded by the Wellcome Trust, which supports our Vital Signs series; funders have no role in Grist’s editorial decisions.) The next phase of Harmonize is to turn the data it has collected at sites in Peru, Colombia, Brazil, and the Dominican Republic into case studies that other researchers can use to determine the extent to which climate change is influencing human health elsewhere in the world.
“The idea is to provide these resources for people in El Salvador or in Nigeria to do their own health attribution studies,” said Ana Maria Vicedo Cabrera, an environmental epidemiologist at the University of Bern in Switzerland, who leads this work. “If we make the tools and the resources more available, the evidence will grow.”
Carrasco-Escobar isn’t the only expert trying to figure out what makes a mosquito tick. On the outskirts of Curitiba, an affluent city in southern Brazil, a squat building on the university campus of the Instituto de Tecnologia do Paraná houses millions of Aedes aegypti. These mosquitoes’ eggs, which are infected with a bacterium called Wolbachia, will be shipped to cities across the country, hatched, and released to mate with wild mosquitoes.
Luciano Andrade Moreira learned about Wolbachia in Australia when he went to visit a researcher named Scott O’Neill in Queensland in 2008. O’Neill’s hypothesis was that the bacteria, which is naturally occurring in fruit flies and many other insects, would shorten the lifespan of mosquitoes. That could give them less time to spread disease. Moreira arrived in Australia in time to see O’Neill make a surprising discovery: The bacteria did something even more powerful — it prevented dengue from replicating inside the insects in the first place. Wolbachia creates a hostile environment for the dengue virus to exist in a mosquito’s gut by priming the insect’s immune system to fight the disease, competing with the virus for cellular resources, and physically crowding out dengue in cells.

Now, almost two decades later, the mosquito factory in Curitiba, which is called Wolbito do Brasil, is teeming with the distant descendants of those first Wolbachia-infected mosquitoes. In the facility, rows of tall machines hold tubes of mosquito eggs. As they hatch into larvae, the insects are fed a mixture of water and protein powder and grow into pupae. Then, the pupae are carted into an adjoining room, where they are washed between two panes of glass — the smaller pupae, the males, slide out of the bottom of the machine first, followed by the larger females. All of the egg-laying females and one of every three males move into yet another room, this one hot and humid, where they are put into mesh enclosures and fed on warm horse blood and sugar water.
On a tour of the factory in November, the facility’s production manager, a biologist named Antônio Brandão, placed his hand next to the mesh and watched hundreds of tiny syringe-like mouths jostle for a better position, as if on cue. “If you come closer and leave your hand there,” he told me, “they will start coming.”
These mesh cages constitute the final step of a mosquito-rearing process that until this point has been accomplished almost entirely by machines, which are carefully supervised by the factory’s 70-odd employees. After the female mosquitoes have laid their eggs on white paper strips at the bottom of the cages, workers will wheel the cages into a refrigerated room and the mosquitoes will finally meet their maker.
Their eggs, however — about 1 million of them collected per cage, which means 100 million eggs are produced every week — are destined for a productive life outside the factory walls. Since the facility commenced operation last summer, six cities in Brazil have started seeding neighborhoods with dengue-fighting mosquitoes, on top of the 10 cities that were already receiving eggs produced by hand by Moreira, Brandão, and other long-suffering employees who worked to popularize the Wolbachia method before the process was automated last year.
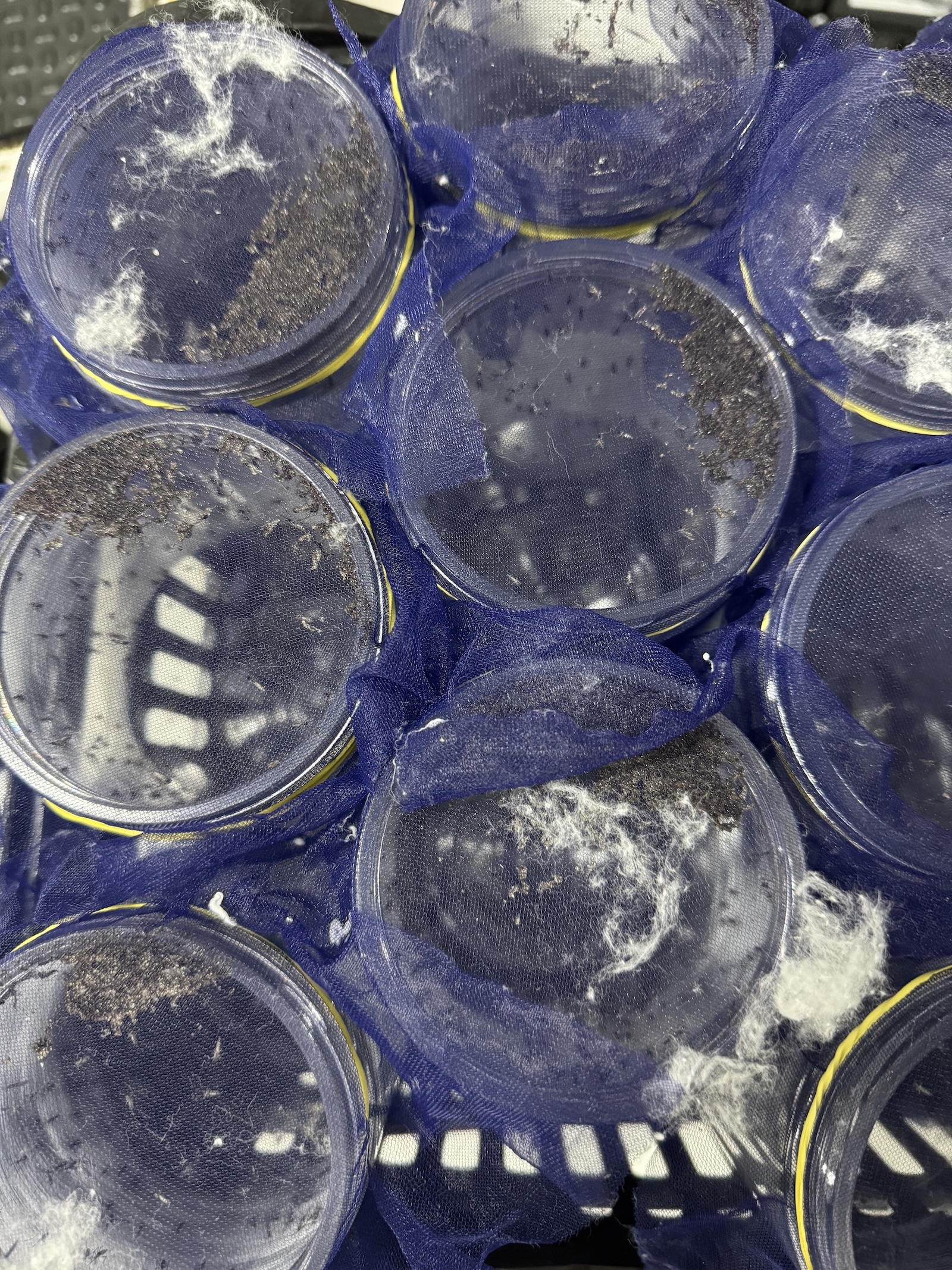

A member of the public health department in Joinville, Brazil, holds up a canister of dengue-fighting mosquitoes. The city in the midst of a campaign to establish Wolbachia in the wild mosquito population. Zoya Teirstein / Grist
Joinville, a two-hour drive from Curitiba, is one of those cities. On an early midweek morning, members of the local health department and Wolbito employees gathered in a distribution center to load a car with canisters of mosquitoes. The mosquitoes were hatched from bags of pellets. Each pellet contains eggs and a feeding mixture, and all participating cities have to do is put the pellets in water and wait for the insects to hatch.
As the car pulled out of the distribution center and wound slowly through Joinville at 7 a.m., Lúcia Jordan, a city health agent, rolled down her window and held an open container in the air, giving it a few hard shakes to eject any lingering mosquitoes. She repeated the exercise over and over as the car made slow, methodical turns through the city’s neighborhoods. A woman waiting for her bus watched the car go by with a bemused expression, seemingly unfazed by the torrent of blood-sucking creatures released in her vicinity.

Much like InnovaLab’s efforts in the Amazon, Wolbito’s work relies heavily on buy-in from the community. But not everyone can be reached, and the releases understandably look suspicious. There are other anti-mosquito initiatives that seek to reduce the overall population of mosquitoes in a given place, but that’s not the goal here. The residents of Joinville will endure no fewer mosquito bites as a result of this program. But, after two years of sustained Wolbachia releases, they will receive widespread and lasting protection from dengue fever, Zika, yellow fever, and other diseases carried by Aedes aegypti mosquitoes.
Niterói, a city of 500,000 connected to Rio de Janeiro, was one of the first places in Brazil to be fully protected by the Wolbachia method. The releases took place from 2017 to 2019. Dengue caseloads declined nearly 90 percent across the city following the treatment, compared to the 10-year average leading up to 2017. During the historic dengue outbreak in 2024, when many Brazilian cities saw record numbers of cases, Niterói recorded fewer than 2,000 — just about a quarter of its pre-treatment average.
“It’s not often you come across a proposal to release mosquitoes when our entire history of disease prevention was to fight against the mosquitoes,” Ana Eppinghaus, health surveillance coordinator at the Municipal Health Foundation of Niterói, said at the time. “We accept the challenge.”
Over the next decade, Moreira aims to protect half of the Brazilian population using Wolbachia. So far, there’s no reason to think he won’t succeed. The method O’Neill pioneered in Australia is being used by 15 countries, and evidence from multiple regions shows dengue incidence drops sharply after Wolbachia mosquitoes establish themselves. Caseloads have fallen in every city in Brazil where Wolbachia has taken hold. Moreira said his biggest problem now is producing mosquitoes fast enough to meet the demand of everyone who wants them.
Despite this, some politicians remain wary of committing to the program — especially when he tells them it might not produce results for a year or more. “Our program is not like a spray that you just spray and kill all the mosquitoes and solve the problem,” he said. “They think it’s too long to wait.”
In the United States, mosquitoes are viewed as a nuisance, rather than the public health disaster they have long been in tropical nations like Brazil. That wasn’t always the case: The Centers for Disease Control and Prevention, or CDC, was established in 1946 to fight malaria around U.S. military bases. It was wildly successful in that mission, all but eradicating the disease from the country by the early 1950s with the help of the devastating chemical DDT. The agency learned a valuable lesson through that effort that still resounds today: Eradicating vector-borne disease is possible “in nations with temperate climates and seasonal malaria transmission.”
But what happens when the climate becomes less temperate? Native and invasive tropical plants and animals move north, as average temperatures rise and winter freezes become weaker. Subtropical states — Florida, Alabama, Mississippi, Louisiana, Texas, New Mexico, Arizona, and California — begin to tropicalize, a process that will be all but complete by the end of the century, according to a 2021 report published by the United States Geological Survey, the Department of the Interior’s science agency.
“Tropical mosquitoes that can transmit encephalitis, West Nile virus, and other diseases,” the report said, “are likely to further expand their ranges, putting millions of people and wildlife species at risk of these diseases.”
Indeed, California saw its first-ever locally-acquired dengue case in October 2023. The California Department of Public Health quickly found another case related to the first — also in someone who had not recently traveled. Cases continued to pop up over the next two years.
In response, the Greater Los Angeles County Vector Control District stepped up its efforts to eradicate mosquitoes. In collaboration with its counterpart in adjacent Orange County, the agency began releasing male mosquitoes sterilized by X-ray, running pilot programs in two neighborhoods in northeast Los Angeles. The relief offered by the campaign is fundamentally temporary: The technique only works as long as the participating counties are committed to regularly releasing mosquitoes. That’s because sterile males produce no offspring, so their genetic lines cannot continue independently. Each new generation of bloodsuckers, in other words, is not necessarily any less deadly than the last.

Other mosquito control efforts in the U.S. in recent years similarly rely on sterilization and continuous releases to work. MosquitoMate, a company based in Kentucky, uses the Wolbachia method — but instead of breeding dengue-resistant insects that can then overtake the native population, as Moreira does in Brazil, MosquitoMate uses the bacteria to naturally sterilize male mosquitoes. This means that it reduces overall populations when the sterilized insects fail to breed, but it’s similar to the X-ray technique in the sense that the sterilized mosquitoes cannot pass on their benevolent quirk to future generations.
The result is just a high-tech variant on the same mosquito suppression paradigm that has been the expected approach in the U.S. since the founding of the CDC. Part of the reason for this stasis is that convenient regulatory frameworks exist for approving mosquito suppression initiatives via the Environmental Protection Agency. Modifying mosquitoes so that they change the characteristics of future generations, on the other hand, involves a much more complicated review process, like the development of biotech products. The legal and ecological ramifications of a Wolbachia-like program would inevitably subject it to yearslong regulatory hoops. So for now, the U.S. is stuck with piecemeal and halfhearted attempts to crush American mosquito populations. The problem is they’re not really working: Aedes populations have exploded in certain hotspots across the country over the past few years.
Recent bipartisan federal legislation would authorize $100 million per year for mosquito surveillance and control, significantly expanding the CDC’s current slate of grants for vector-borne disease monitoring and preparedness. The bill, introduced in 2023, seeks to stitch together the various fragmented and underfunded mosquito control efforts taking place across the country into a more cohesive framework. But the “SMASH Act,” short for Strengthening Mosquito Abatement for Safety and Health (a reauthorization of a weaker SMASH program created in 2019), has stalled out in a legislative committee.
Congress may be in no hurry, but time is not on its side. If the country’s climate, long one of its greatest allies in the fight against malaria and other vector-borne maladies, is changing, then the U.S. is due for an overhaul of its relationship with tropical illnesses. The path forward may require developing systems to catch the clues that herald a disease outbreak, as Carrasco-Escobar is doing in Peru. Or valuing long-term disease suppression over fewer mosquitoes in the short-term — Moreira’s goal in Brazil. Or maybe it’s some combination of the two — the double-tipped spear that seeks to work with the natural world rather than against it.
But first, Durand told me at the hospital in Lima, temperate countries need to recognize that the ground beneath them is shifting. When he looks at California, he sees echoes of Lima’s 2024 outbreak.
“That’s how we started,” he said. “With local cases.”








